V oblasti elektrochemické analýzy nukleových kyselin má OBCMO světové prvenství díky objevu elektroaktivity DNA, který uskutečnil prof. Emil Paleček koncem 50. let [Naturwiss. 45 (1958), 186-187, Nature 188 (1960) 656-657]. Tento objev ukázal, že je možné pozorovat signály adeninu, cytosinu a guaninu pomocí oscilografické polarografie na kapající rtuťové elektrodě. Od té doby se oddělení intenzivně zabývá studiem chování nukleových kyselin na elektrodách a v roztocích, což vyústilo v položení základů oboru elektrochemie nukleových kyselin.
Po počátečním období, kdy se při analýze DNA používala pouze rtuťová elektroda, v 70. letech ukázali Viktor Brabec a Glenn Dryhurst [Electroanal. Chem. 89 (1978), 161-173, Electroanal. Chem. 89 (1978), 161–173, Bioelektrochemie Bioenerg. 8 (1981), 437–449], že DNA (báze adenin a guanin) poskytuje analyticky využitelné signály také na uhlíkových elektrodách. Dalšího rozvoje v oblasti elektrochemie nukleových kyselin bylo dosaženo zavedením adsorpční přenosové rozpouštěcí metody, která vedla ke snížení objemu vzorku potřebného pro analýzu a ke zvýšení citlivosti [J. Electroanal. Chem. 214 (1986), 359-371].
Mezi další oblasti elektrochemie nukleových kyselin, na jejichž rozvoji se DBCMO značnou měrou podílelo, patří:
- vývoj technik pro detekci struktury DNA
- studium elektrochemického chování poškozené DNA a vývoj senzorů pro detekci poškození DNA
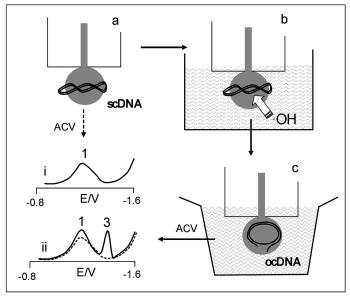
- vývoj elektrochemických technik pro detekci hybridizace DNA
- systematické studie peptidové nukleové kyseliny (PNA)
- značení nukleových kyselin elektrochemicky aktivními skupinami. Komplexy oxidu osmičelého byly prvními značkami použitými pro elektrochemické značení DNA, následovala řada dalších značek DNA.
- zavedení a aplikace "dvoupovrchových" elektrochemických technik (s využitím magnetických kuliček) při studiu hybridizace DNA a interakcí DNA-protein
- studium elektrochemického a mezifázového chování syntetických oligonukleotidů (ODN) s ohledem na obsah nukleobází a sekvenci nukleotidů; studium interakcí DNA, ODN a složek DNA s různými povrchy elektrod
Mezi nejnovější příspěvky oddělení v oblasti elektrochemie nukleových kyselin patří:
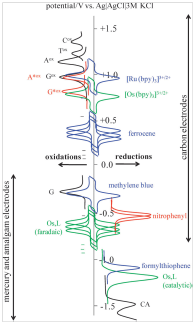
- uhlíkové elektrody, zejména elektroda z pyrolytického grafitu s bazální orientací, jsou vhodné pro studium nejen anodických oxidačních procesů, ale i katodické redukce nukleobází až do potenciálu -2,0 V [Electrochem. commun. 82 (2017), 34–38].
- DNA je schopna katalyzovat vylučování vodíku, což lze pozorovat jako elektrokatalytický pík pomocí voltametrie nebo chronopotenciometrie na rtuťových a pevných amalgámových elektrodách [ChemElectroChem 5 (2018), 936–942]
Další oblastí výzkumu, do které OBCMO přispívá, je elektrochemie proteinů a nové metody pro analýzu proteinů. To bylo umožněno objevem tzv. „peaku H“ [Electroanalysis 10 (1998), 403–409], který za určitých podmínek poskytují peptidy a proteiny na rtuťových elektrodách s využitím chronopotenciometrické analýzy s konstantním proudem. Tento pík lze pozorovat díky schopnosti proteinů a peptidů katalyzovat vylučování vodíku. Peak H umožňuje nejen vysoce citlivou detekci proteinů, včetně těch důležitých v biomedicíně a výzkumu rakoviny, ale také specificky reaguje na strukturní změny proteinů, jako je denaturace, agregace, výměna aminokyselin atd. Tento přístup otevřel nové možnosti aplikace elektrochemických metod pro studium protein-protein [Electrochim. Acta 297 (2019), 974–979] a/nebo protein-DNA interakcí [Chem. Rev. 115 (2015), 2045–2108].
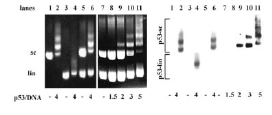
V oblasti studia nádorového supresoru p53 má DBCMO dlouhou tradici. V 90. letech 20. století byl objeven nový typ interakce proteinu p53 s DNA – preferenční vazba p53 na superhelikální (sc) DNA [Oncogene 15 (1997), 2201–2209]. Bylo zjištěno, že p53 se váže na scDNA neobsahující specifické vazebné místo pro p53 s mnohem vyšší afinitou než na lineární DNA obsahující tuto nukleotidovou sekvenci. Další studie ukázaly, že C-terminální doména proteinu p53 a oligomerní stav jsou pro tento jev klíčové a že superhelicita DNA ovlivňuje sekvenčně specifickou vazbu p53 na jeho cílová místa.